Showing 1 to 15 of 2504 results
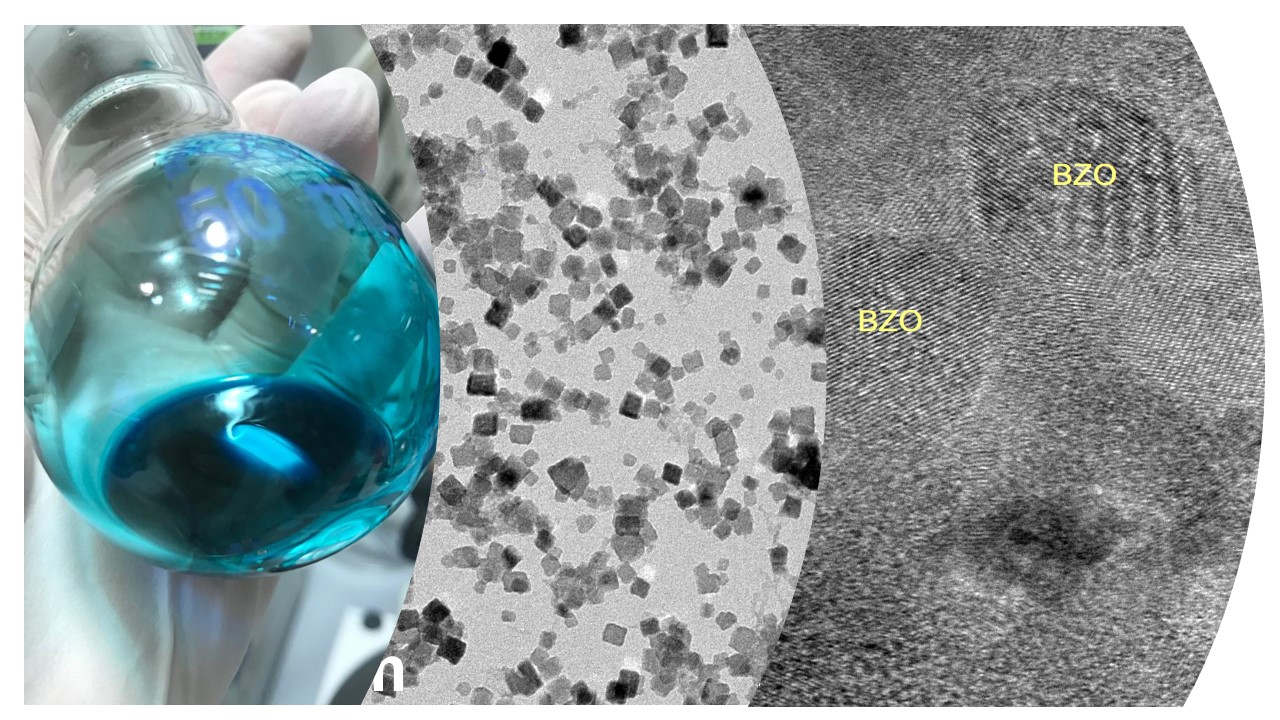

Ink for the production of superconducting flexible tapes
Patents for licensing CSIC - Consejo Superior de Investigaciones Científicas

FIBRACEP: New dietary fibre-based ingredient to control cardiovascular disease risk
Innovative Products and Technologies Universitat de Lleida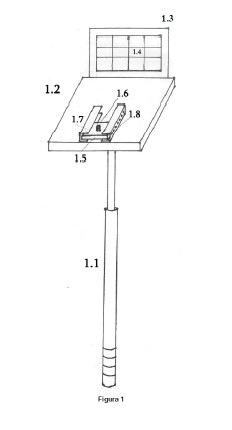

Device for taking pictures of the treetops
Patents for licensing Universidad de Alcalá-OTRI

Wide-area surveillance system
Innovative Products and Technologies CSIR

Energy harvesting from ambient vibrations for wireless sensors application
Patents for licensing Innoget Member

New active compound for the prevention and treatment of infections caused by Zika virus (ZIKV)
Patents for licensing Centre Technology Transfer CITTRU

US9369082 (B2) - Solar power mobile charging station
Patents for licensing Syncoda Technologies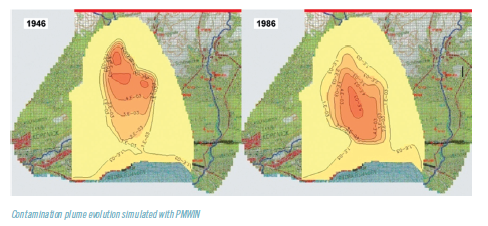

Water resources and contaminant transport modelling
Innovative Products and Technologies IMDEA Water Institute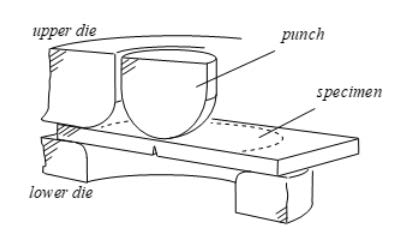

Evaluation of mechanical and fracture properties by miniature punching test
Research Services and Capabilities UNIVERSIDAD DE BURGOS

New test for components subjected to high pressure (up to 6000 bar)
Research Services and Capabilities UNIVERSIDAD DE BURGOS

mynd, a powerful AI-driven text mining platform
Innovative Products and Technologies mynd

A propulsion/drive system of aquatic vessels.
Patents for licensing Cracow University of Technology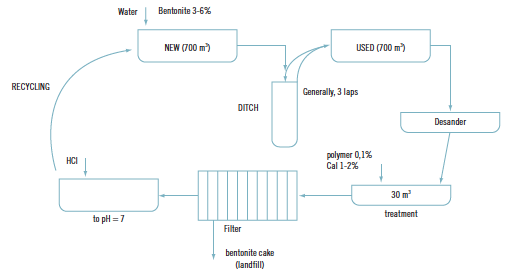

Treatment of water from bentonite slurry
Patents for licensing IMDEA Water Institute

Education system for 3D drawing chemical structures
Innovative Products and Technologies Centre Technology Transfer CITTRU