Showing 1 to 15 of 2456 results


Innovative heat-reflective coating for indoor application
Innovative Products and Technologies Laser Consult Ltd.

Combined Determination of the Concentration and Enantiomeric Composition of Chiral Compounds Using Single Chiroptical Assay
Innovative Products and Technologies Georgetown University

Rich Fertilize in Siderophore, that allows to cure iron chlorosis
Patents for licensing Universidad Adolfo Ibáñez
LSF: B2B Chemtech, UK & NL, Angel round with lead investor, ≈40% funding secured
Investment Opportunities in Startups and Spinoffs Low Sulphur Fuels Ltd

The new lupeol derivative and the method of obtaining.
Innovative Products and Technologies Cracow University of Technology

Soy-Based Corrosion Inhibitor for Ferrous Metals
Innovative Products and Technologies Airable Research Lab, business line of Ohio Soybean Council

Tethered Aerostat
Innovative Products and Technologies CSIR

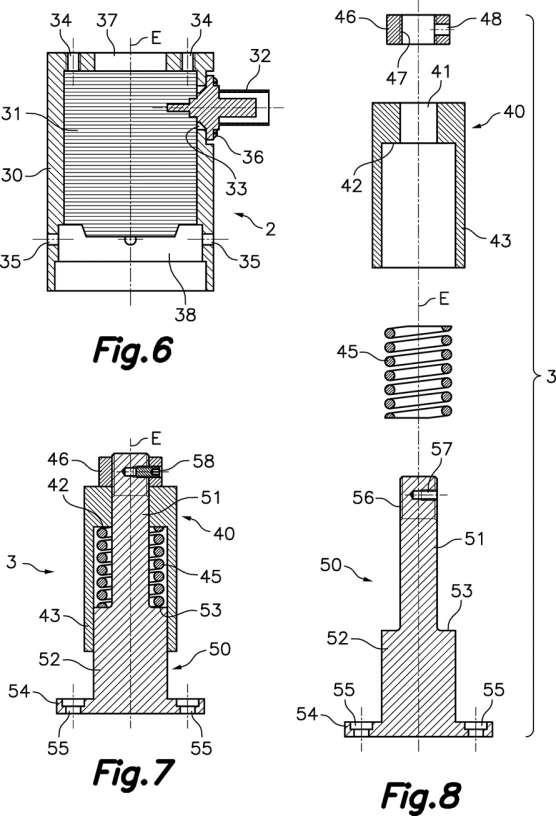
DEVICE FOR SPERM SELECTION BY THERMOTAXIS
Patents for licensing INIA

Conductive cellulose nanocrystals
Innovative Products and Technologies University of Waterloo

Curriculum for Non-Clinical Ophthalmic Medical Assisting
Innovative Products and Technologies Georgetown University

Process for the photocatalytic oxidation of alkanes and aromatic hydrocarbons
Innovative Products and Technologies Universitat de Girona

Pc-protec additive
Patents for licensing Universitat Politècnica de Catalunya - UPC

Monitoring of Esca viticulture disease symptoms
Innovative Products and Technologies Luxembourg Institute of Science and Technology (LIST)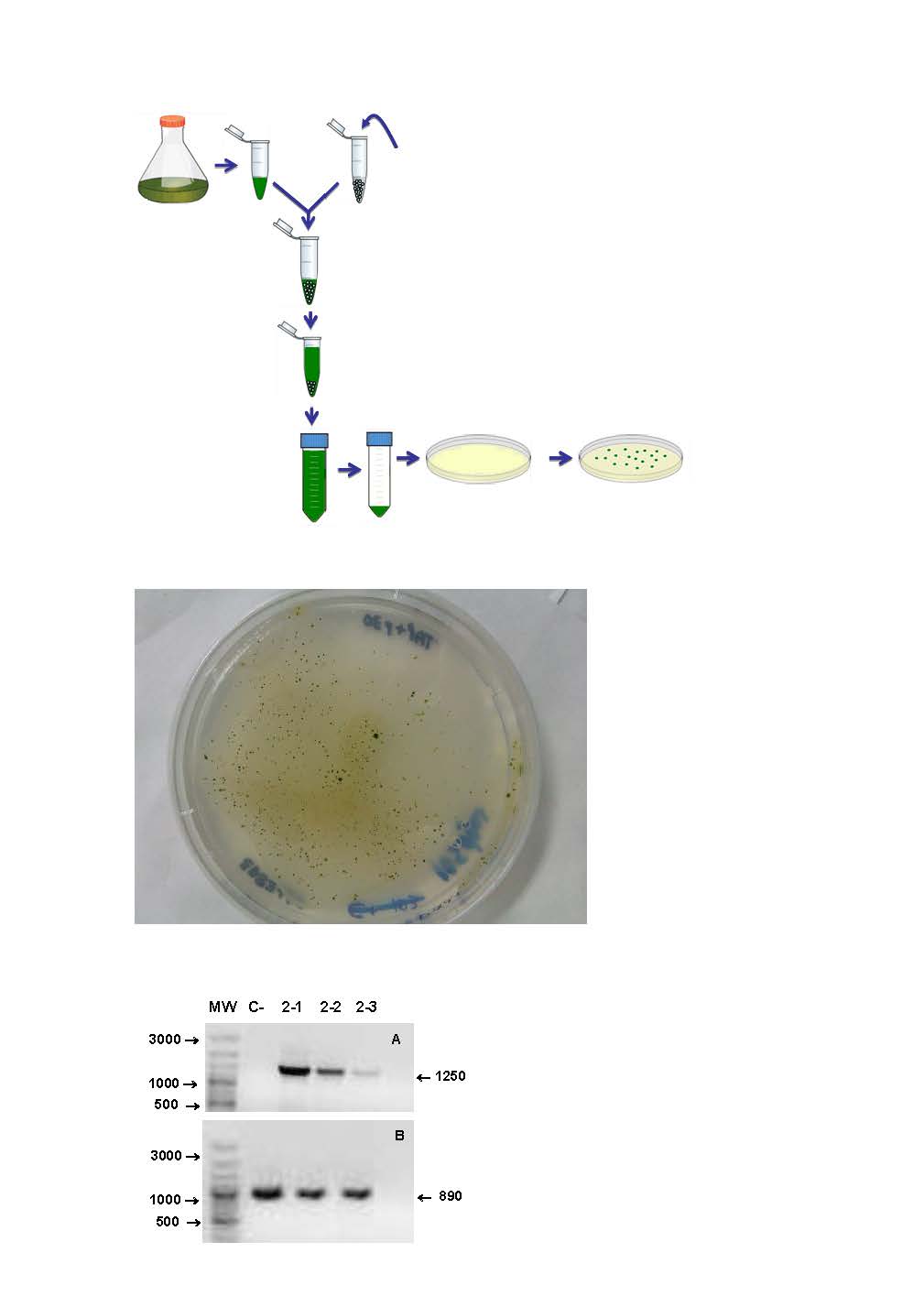

New procedure for the genetic transformation of plant cells and kit for carrying out said method
Knowhow and Research output University of Huelva