Showing 1 to 15 of 2076 results


Predict method of Pregnant Gestational age
Innovative Products and Technologies Fundación para la Investigación Biosanitaria de Andalucía Oriental (FIBAO)

Biomarkers to predict response to treatment in metastatic melanoma patients
Patents for licensing IBIMA-Plataforma Bionand

Rice husk as a filter for the removal of contaminants in water
Patents for licensing Consejo Superior de Investigaciones Científicas

Precision arm for fixing a surface antena in a sample in magnetic resonance
Patents for licensing CSIC - Consejo Superior de Investigaciones Científicas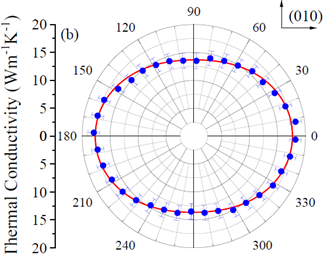

new contactless method for the measurement of thermal conductivity in materials
Patents for licensing ICMAB-CSIC

Method for preparing advanced Eco-friendly incombustible biocomposite
Innovative Products and Technologies Korea Institute of Energy Research

Utilization of Waste Plastic in Designing tiles for Societal Applications
Innovative Products and Technologies National Physical Laboratory

Label Free Detection and Imaging of Biofilms
Knowhow and Research output National Biofilms Innovation Centre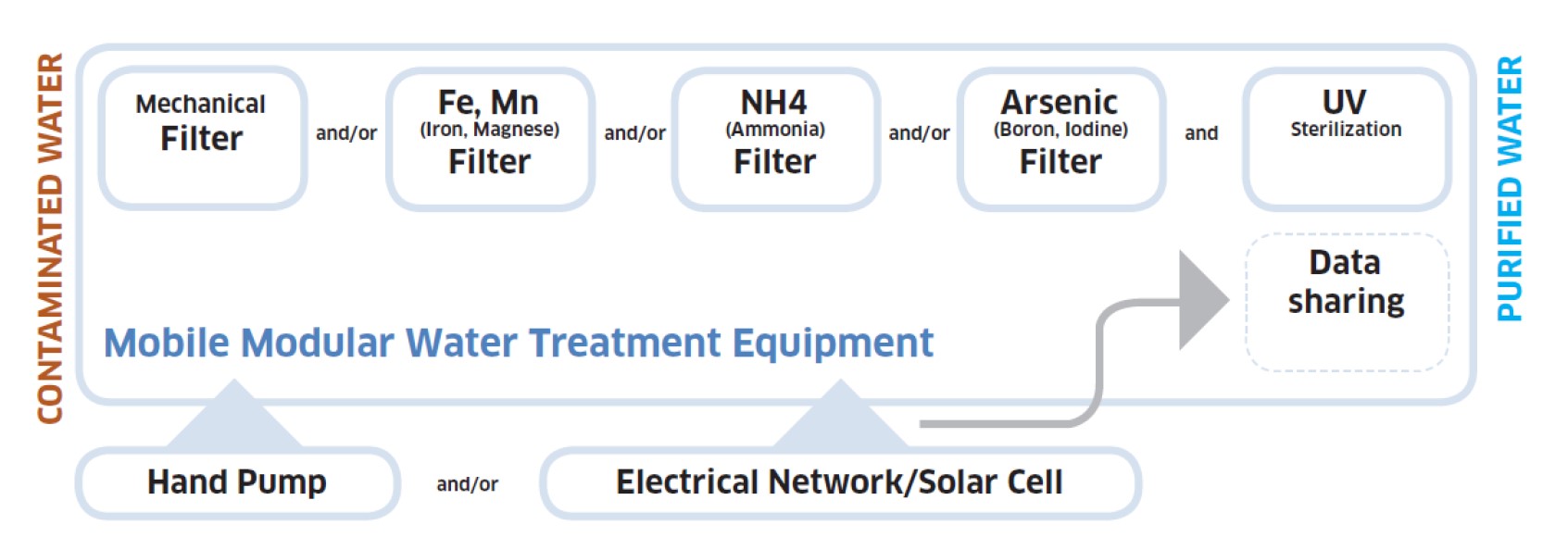

Chemical Free Regenerable Water Filtering System for Small Communities with Contaminated Water
Innovative Products and Technologies Laser Consult Ltd.

Method for determining prognosis in subjects diagnosed with pulmonary arterial hypertension
Patents for licensing CINBIO

Point of Use Decentralized Delivery Fresh Drinking Water Daily via Atmospheric Water Generators
Innovative Products and Technologies Green Technology Global, Inc.

Laboratory for Trace Organic Analyses - Cracow Univerisity of Technology
Research Services and Capabilities Cracow University of Technology

Preparation method of palladium alloy composite membrane for hydrogen separation.
Innovative Products and Technologies Korea Institute of Energy Research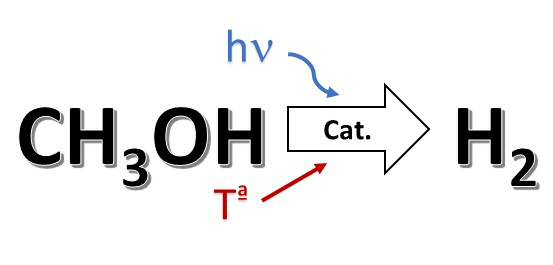

Method for hydrogen production by photo-thermal catalysis
Patents for licensing CSIC - Consejo Superior de Investigaciones Científicas