Showing 1 to 15 of 2456 results
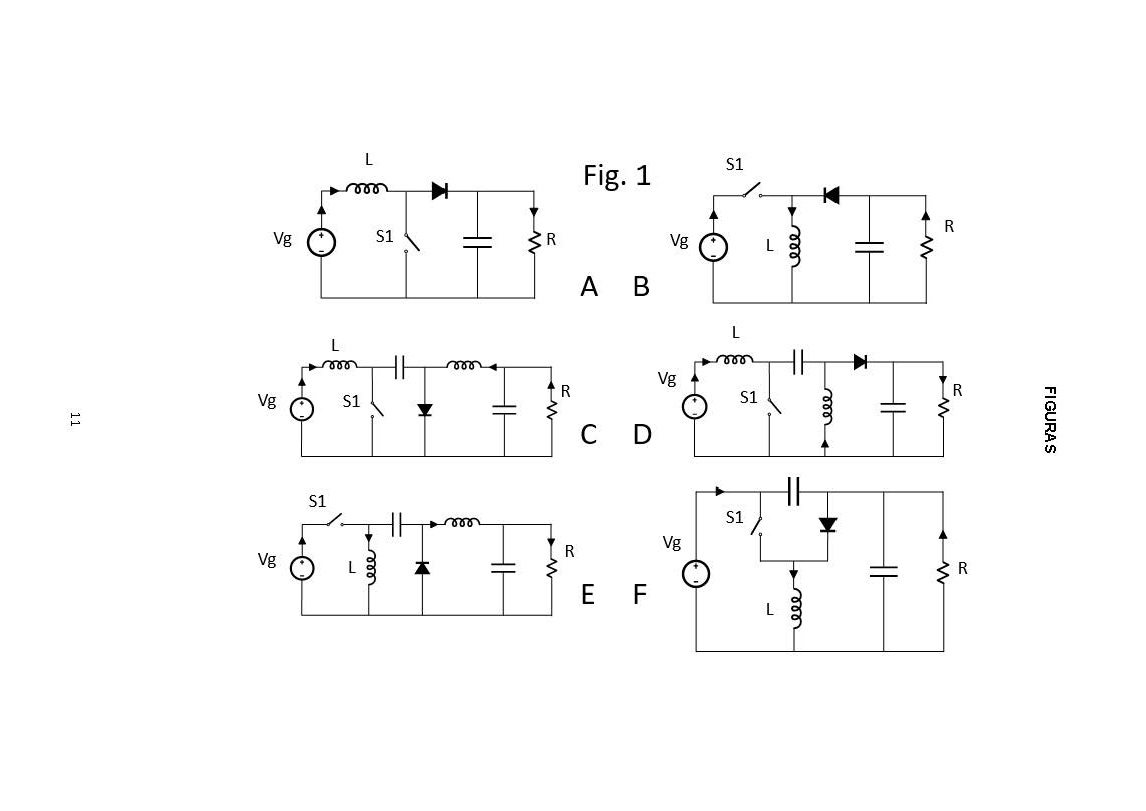

NEW CONFIGURATIONS OF CC-CC CONVERTERS OF ONE INPUT AND MULTIPLE OUTPUTS WITHOUT TRANSFORMER AND POWER CONVERTER THAT APPLY THEM
Patents for licensing University of Huelva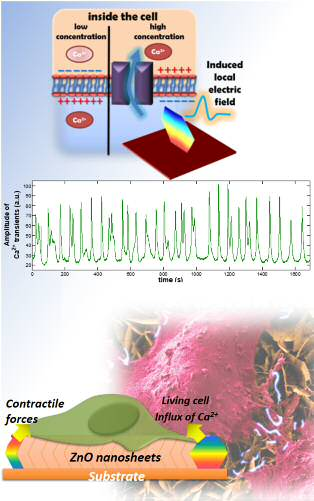

Biocompatible self-generating cell stimulator
Patents for licensing Consejo Superior de Investigaciones Científicas

Alpha Lifetech-Phage Display For Antibody Discovery
Research Services and Capabilities Alpha Lifetech Inc.

Granular Biomass: Method for Wastewater Treatment
Innovative Products and Technologies Universidad Adolfo Ibáñez

EmDrive two is new version of this machine, which provides huge thrust, big enough for satelites and flying vehicles.
Knowhow and Research output private researcher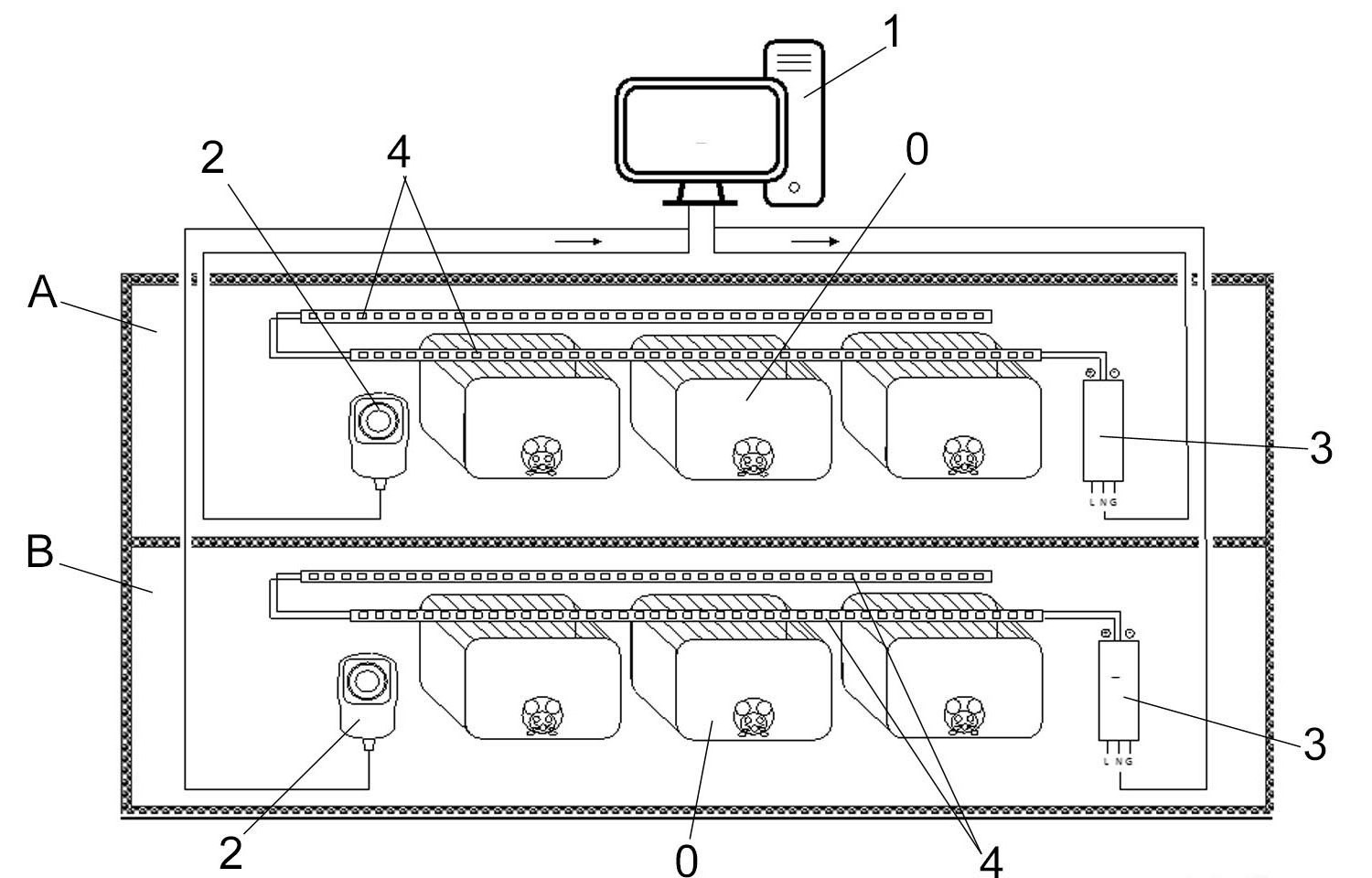

Novel individualized, homogeneous and adjustable lighting system for animals
Patents for licensing Universidad de Alicante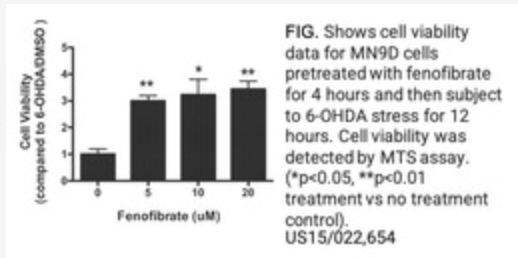

Repositioning Fenofibrate for the Treatment of Neurodegenerative Disease
Innovative Products and Technologies Georgetown University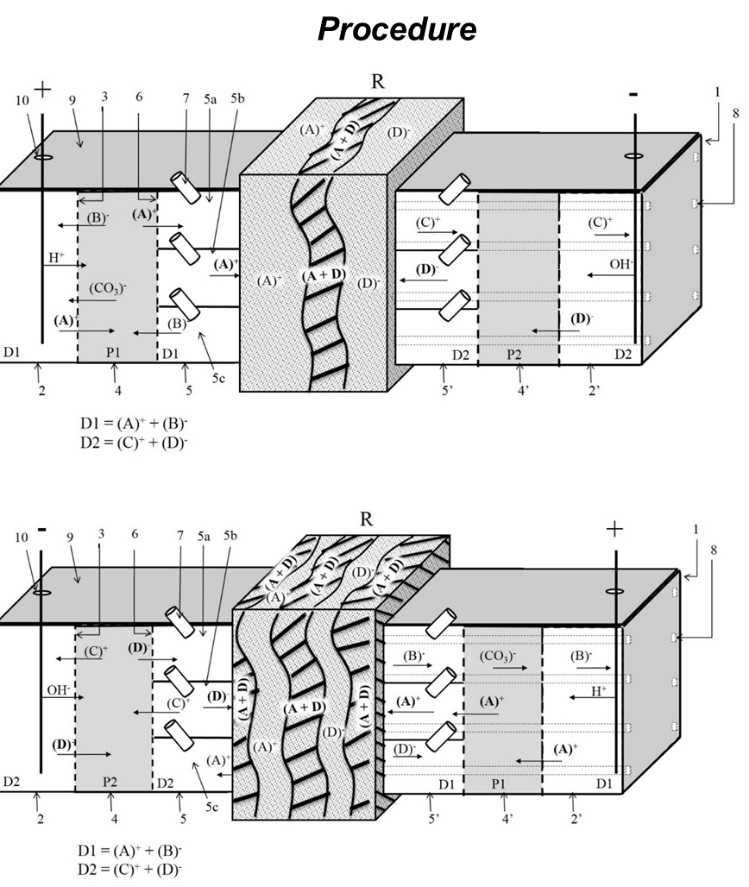

Electrokinetic device and procedure for consolidation of porous materials
Patents for licensing University of Vigo

Speech Gap Test – novel automatic speech recognition of Mild Cognitive Impairment
Patents for licensing University of Szeged![A new method of obtaining mixoxanthophylls from Arthrospira platensis dye extracts (from spirulina) for use in the pharmaceutical […]](https://static6.innoget.com/uploads/88e3c8b21489c0f2a98efd73be95b10d4e5f691f.jpg)

A new method of obtaining mixoxanthophylls from Arthrospira platensis dye extracts (from spirulina) for use in the pharmaceutical […]
Patents for licensing Jagiellonian University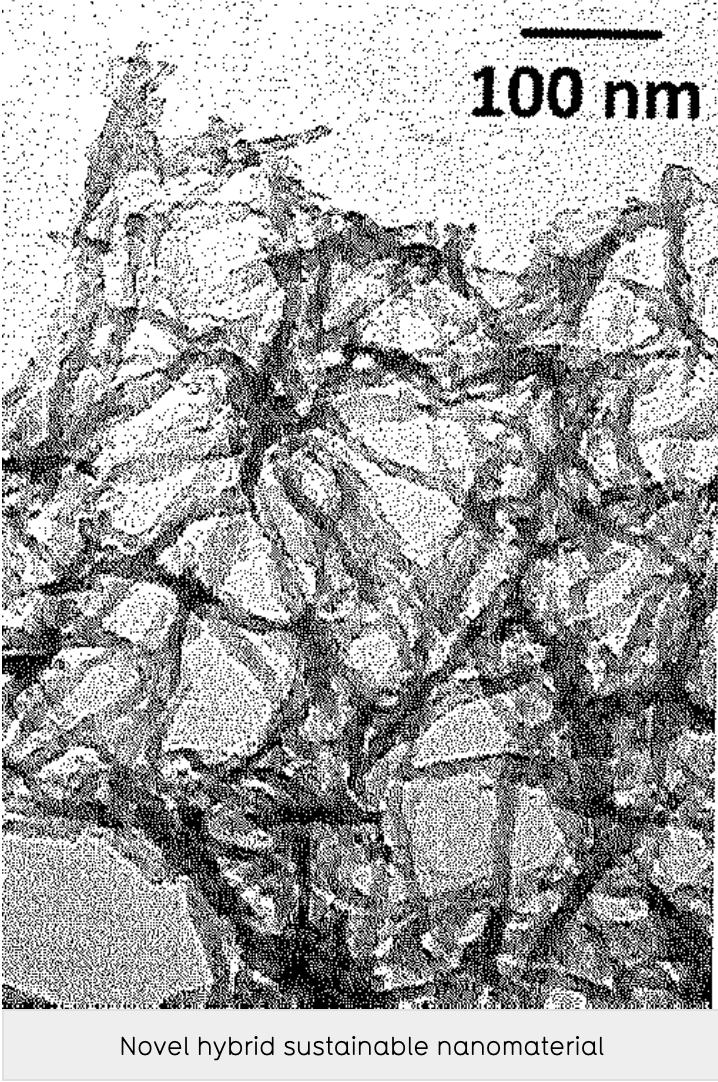

Process to prepare amine-functionalized cellulose nanocrystals for applications such as an antimicrobial agent or catalyst
Innovative Products and Technologies University of Waterloo

Augmented Reality on Android: Arquiz
Innovative Products and Technologies UNIVERSIDAD DE BURGOS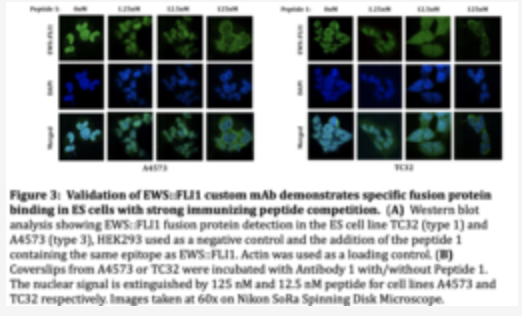

Custom FLI I Monoclonal Antibody
Innovative Products and Technologies Georgetown University

Low energy building design and thermal refurbishment of existing buildings
Research Services and Capabilities Cracow University of Technology

