Showing 1 to 15 of 2455 results

Creation of personalized tourist routes for android devices
Patents for licensing UNIVERSIDAD DE BURGOS

Fire Retardant Apparatus (FRA): Enhancing Climate Change Adaptation Through Optimized Wildfire Management
Innovative Products and Technologies Universidad Adolfo Ibáñez

Claw hand rehabilitation device and procedure for use
Innovative Products and Technologies UNIVERSIDAD DE BURGOS

Universal sensor platform for immunoassay
Patents for licensing Universidad de Alcalá-OTRI

Rich Fertilize in Siderophore, that allows to cure iron chlorosis
Patents for licensing Universidad Adolfo Ibáñez

Hepatitis B Virus-Woodchuck Hepatitis Virus Chimera for Hepatitis B Virus Infection of Woodchucks
Innovative Products and Technologies Georgetown University

Bio-Based Broad-Spectrum Herbicide
Innovative Products and Technologies Airable Research Lab, business line of Ohio Soybean Council

Cmore platform
Innovative Products and Technologies CSIR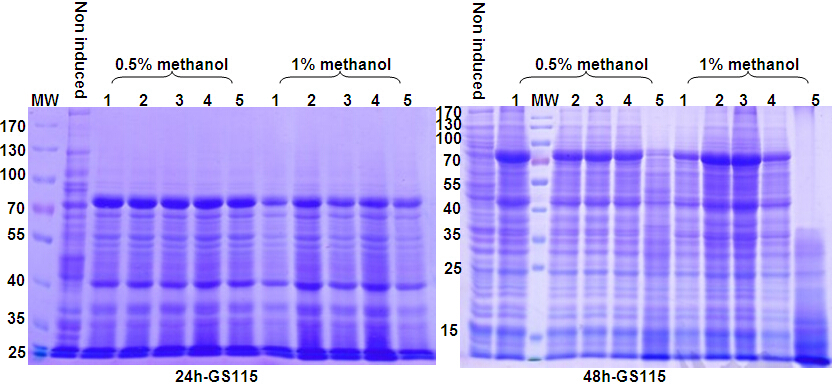

Yeast Expression System Service
Research Services and Capabilities Leading Biology

Vehicle rollover control using pulsed active steering control strategy
Innovative Products and Technologies University of Waterloo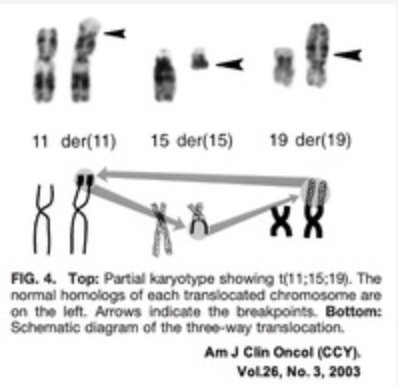

NUT midline carcinoma cell lines TC797 and TC1297
Innovative Products and Technologies Georgetown University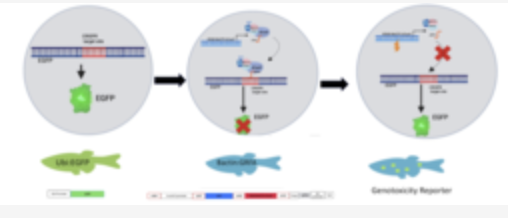

Real-time zebrafish reporter assay for monitoring tissue-specific genotoxicity
Innovative Products and Technologies Georgetown University![Bioremediation strategy in situ for the precipitation and immobilization of uranium and other toxic elements in contaminated soils[…]](https://static7.innoget.com/uploads/f396de9edf618ff5c2e02cbc0b1944f438af1c89.png)

Bioremediation strategy in situ for the precipitation and immobilization of uranium and other toxic elements in contaminated soils[…]
Patents for licensing Universidad de Alcalá-OTRI

Sustainable gypsum prefabricated from polyurethane foam residue
Patents for licensing UNIVERSIDAD DE BURGOS

