Showing 1 to 15 of 2456 results


Laboratory-scale photoreactors for high-througput experimentation
Innovative Products and Technologies Institute of Chemical Research of Catalonia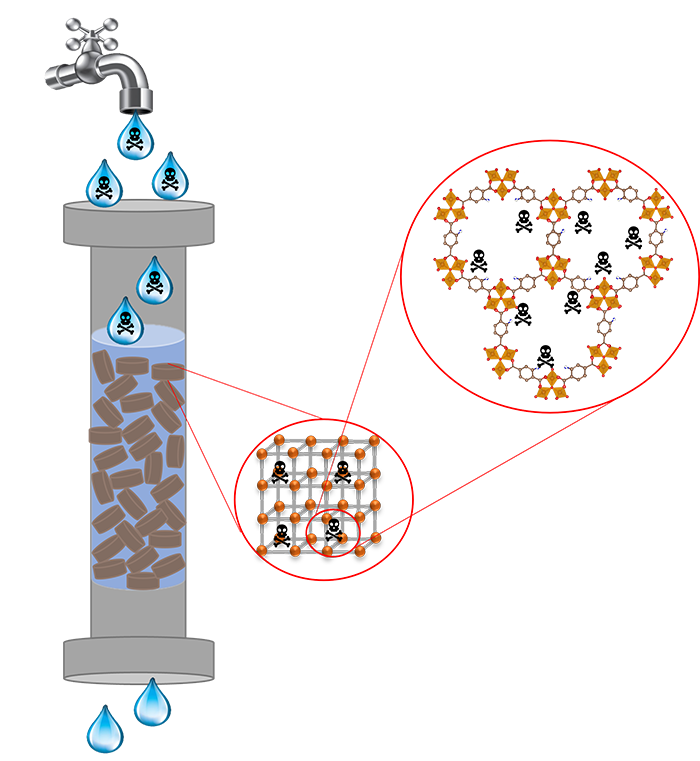

Water remediation using novel advanced porous materials
Innovative Products and Technologies IMDEA Energy

Antimalarial Agent, Methods and Uses Thereof
Patents for licensing NOVA University Lisbon

BTS-PWR. Equipment for biogas line in WWTP. Removal particle and moist in biogas stream
Innovative Products and Technologies Biogas & Gases Technologies

High Voltage Photovoltaic Cells with High Efficiency
Innovative Products and Technologies RAMOT at Tel Aviv University Ltd.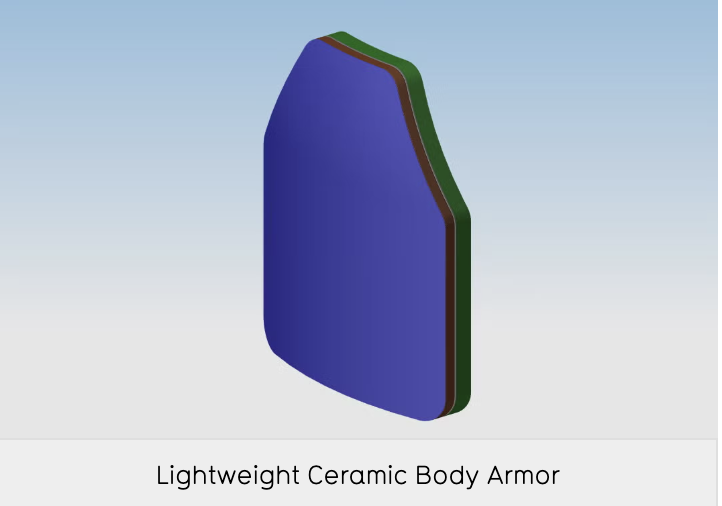

Lightweight ceramic body armor
Innovative Products and Technologies University of Waterloo

ParametricAI: A predictive analytics software
Innovative Products and Technologies University of Waterloo

New enviromental friendly method for the catalytic reduction of sulfoxides using poliols (pinacol or glycerol) as reducing agents
Innovative Products and Technologies UNIVERSIDAD DE BURGOS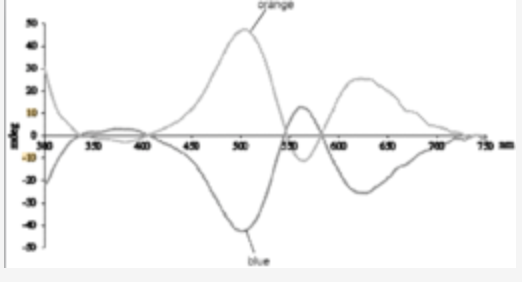

Quantitative Auxiliary-free Chirality Sensing with a Metal Probe
Innovative Products and Technologies Georgetown University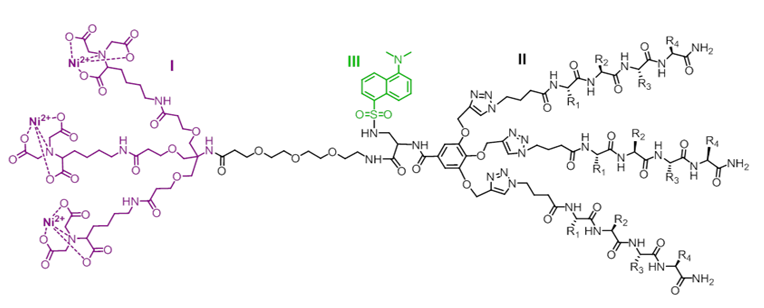

Highly Selective Fluorescent Biosensor for Labeling His-Tagged Proteins
Patents for licensing Yeda

Micro-Enterprise Media Engine (MEME)
Innovative Products and Technologies CSIR

CHEMIOTHERAPY: IMATINIB DRUG MONITORING
Patents for licensing National Cancer Institute CRO Aviano

Innovative portable LAMP-based Technology for On-Site pathogen detection.
Innovative Products and Technologies University of Galway

Market research, consumer studies and advice for the development of online and mobile commerce
Innovative Products and Technologies Universidad de Burgos

