Showing 1 to 15 of 2075 results
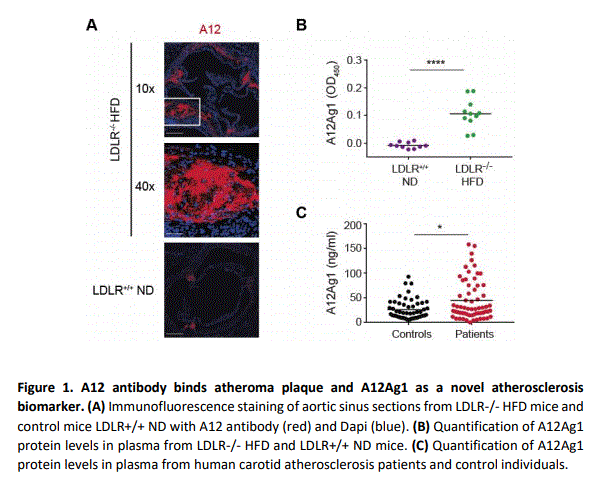

Antibodies for the diagnosis and/or treatment of atherosclerosis
Patents for licensing Spanish National Center for Cardiovascular Research (CNIC)

A new method of removing cells (decellularization) from adipose tissue and the use of the obtained preparation in medicine
Patents for licensing Jagiellonian University

Polymeric membranes with antiseptic capacity for food and textile aplicattion
Patents for licensing UNIVERSIDAD DE BURGOS

ORAL STREPTOCOCCI WITH BACTERICIDAL ACTIVITY
Patents for licensing Fundación Biomedica Galicia Sur

MATCHX - End-to-end deep coverage network-solution
Innovative Products and Technologies EIT Digital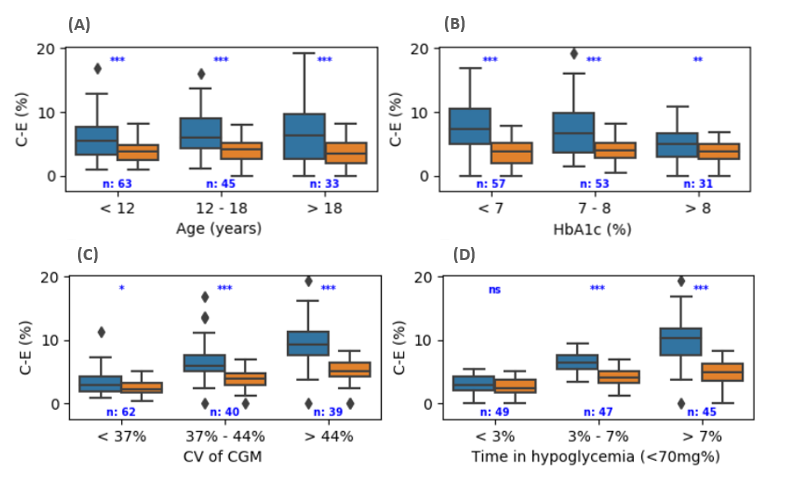

Artificial Pancreas
Patents for licensing Yeda

Speciment for characterization tests under direct tension of concrete and other materials
Patents for licensing UNIVERSIDAD DE BURGOS

New solution to prevent excessive wear in gear, useful for wind turbines
Patents for licensing Universitat Politècnica de Catalunya - UPC

WEBIO - AI-driven Customer Engagement via Conversational Messaging
Innovative Products and Technologies EIT Digital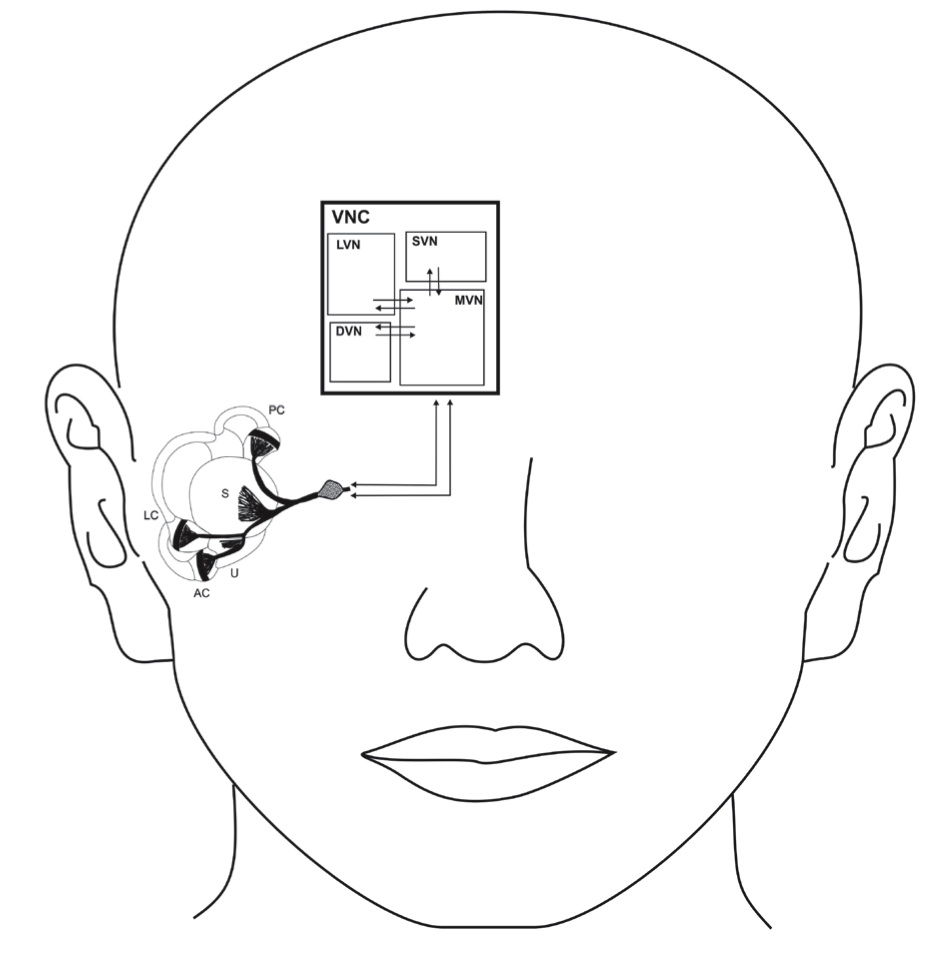

Vestibular prosthesis with micro-electric-mechanical sensors
Patents for licensing Benemérita Universidad Autónoma de Puebla

Real Time Water Quality Monitoring Technology
Patents for licensing Newcastle University

IN VITRO METHOD FOR THE DIAGNOSIS OR PROGNOSIS OF NEURODEGENERATIVE DISORDERS
Patents for licensing Fundación Biomedica Galicia Sur

EIRATECH ROBOTICS - Goods-to-person robotics solution
Innovative Products and Technologies EIT Digital

Non-invasive detection of scuticociliatosis in flat fishes
Patents for licensing CSIC - Consejo Superior de Investigaciones Científicas

