Showing 1 to 15 of 2075 results


Discoloration of effluents by membrane technologies
Research Services and Capabilities UNIVERSIDAD DE BURGOS

Extraction of bioactive compounds from plants
Patents for licensing UNIVERSIDAD DE BURGOS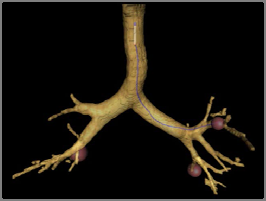

Motion Planning Procedure for the Virtual Bronchoscopy
Patents for licensing Universitat Politècnica de Catalunya - UPC

Stackable container for selective collection of glass bottles
Patents for licensing Universitat Politècnica de Catalunya - UPC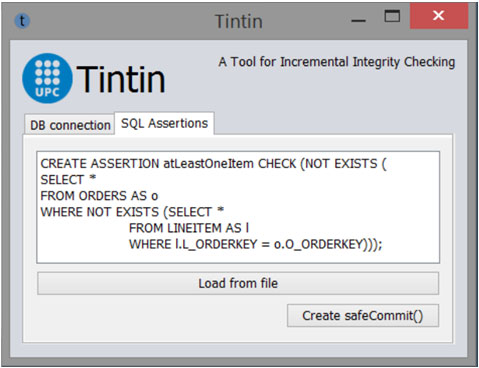

TINTIN: A Tool for INcremental INTegrity checking
Patents for licensing Universitat Politècnica de Catalunya - UPC

Design of training programs and learning resources that promote autonomy and prevent dependence onold people.
Research Services and Capabilities UNIVERSIDAD DE BURGOS

Odor Detection Platform - Low power, Low cost, Programmable Sensor Array
Innovative Products and Technologies University of Manitoba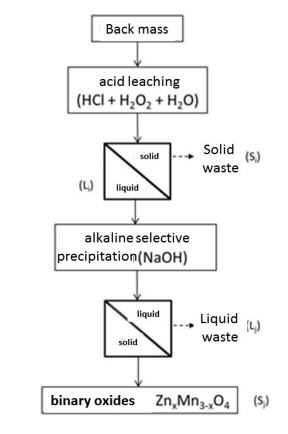

Biocatalysts for the food, pharmaceutical and cosmetic industry made from used batteries
Patents for licensing Consejo Superior de Investigaciones Científicas

Novel Pseudomonas Strain for the Treatment of Iron-Deficient Soil
Innovative Products and Technologies Hub APTA

Neural models based on unsupervised learning for the visualization of large data sets
Research Services and Capabilities UNIVERSIDAD DE BURGOS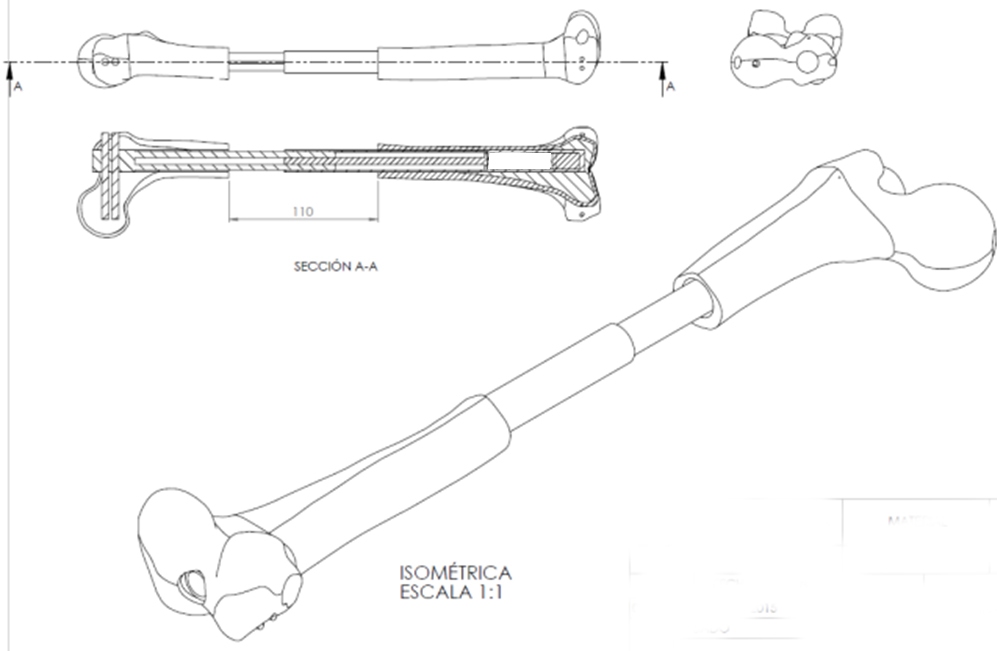

Self-supporting and autonomous prosthesis for the progressive lengthening of long bones
Patents for licensing UNIVERSIDAD DE BURGOS

One-step water recycling for inorganic and organic pollutants
Patents for licensing Universitat de València

Syrupy products powder preparation method
Patents for licensing UNIVERSIDAD DE BURGOS

MUST - Managing Urban Spaces Together
Innovative Products and Technologies Luxembourg Institute of Science and Technology (LIST)

