Showing 1 to 15 of 2075 results
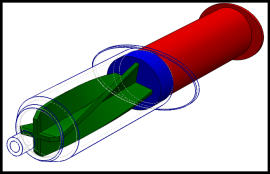

Novel syringes and concepts for homogeneous injection of bone cements
Patents for licensing Universitat Politècnica de Catalunya - UPC

Nitroxoline for prevention and treatment of coronavirus disease
Patents for licensing University of Ljubljana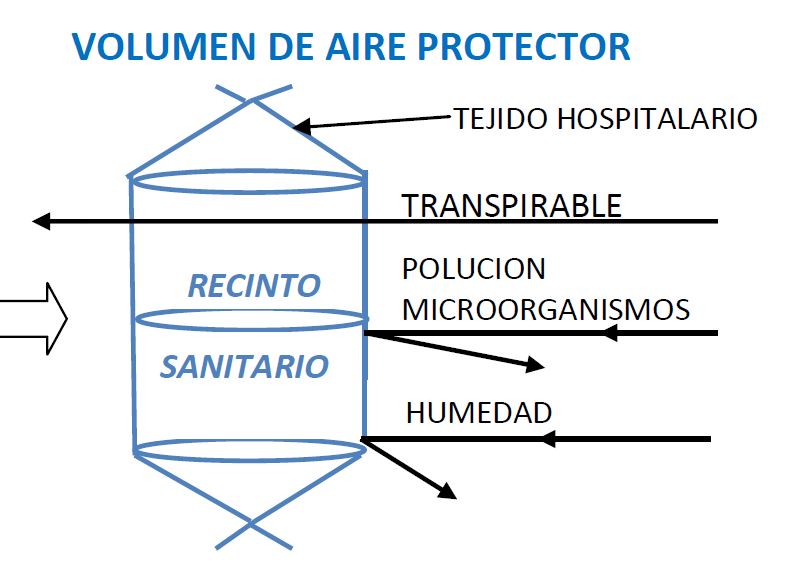

Volumetric protector of burned or wounded extremities
Patents for licensing Asgard Consulting

Producing bioethanol and/or glucose syrups from raw starch
Patents for licensing Unismart - University of Padua Foundation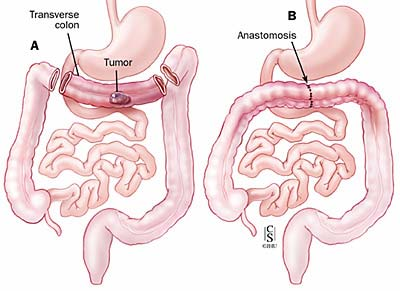

Automatic device for suture in anastomosis surgery interventions
Patents for licensing Universitat Politècnica de Catalunya - UPC

GEO-MOBILITY: Application that helps people with disabilities or reduced mobility to control their environment
Patents for licensing UNIVERSIDAD DE BURGOS

Colorimetric sensor array for pH measurement
Patents for licensing Unismart - University of Padua Foundation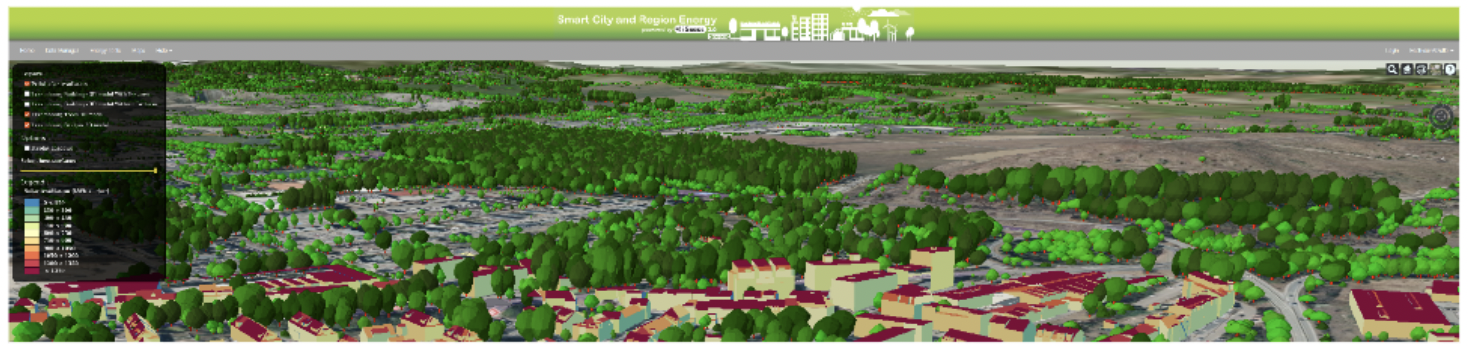

iGuess4SECAP - An Energy Transition Platform for Sustainable Energy and Climate Action Planning for Cities and Regions
Innovative Products and Technologies Luxembourg Institute of Science and Technology (LIST)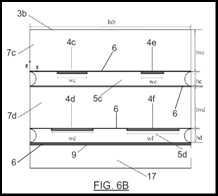

Reconfigurable beam reflectarray antenna for frequencies in the range of terahertz and millimeter wave
Patents for licensing Universitat Politècnica de Catalunya - UPC

Water and soil laboratory with accreditation ISO 17025 EN
Research Services and Capabilities IMDEA Water Institute

Poly divinylbenzene for polypeptide synthesis
Patents for licensing Unismart - University of Padua Foundation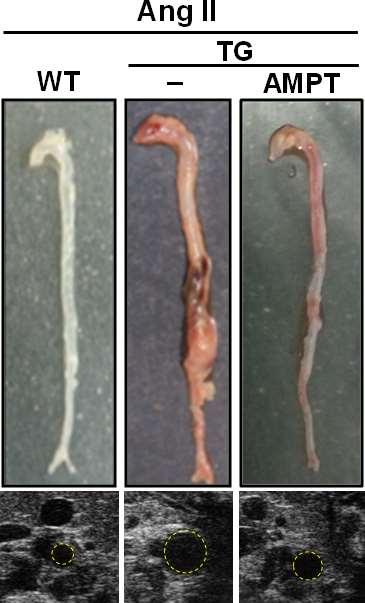

Tyrosine hydroxylase inhibitors for treatment of aortic aneurysm
Patents for licensing CSIC - Consejo Superior de Investigaciones Científicas

New prebiotics from biomass polysaccharides (hemicelluloses and pectins) for food and feed
Patents for licensing University of Vigo

Scalable burst assembly algorithm for Optical Burst Switching networks
Patents for licensing Universitat Politècnica de Catalunya - UPC

