Showing 1 to 15 of 2075 results


Photobiologically friendly phosphor converted light-emitting diode
Innovative Products and Technologies Vilnius University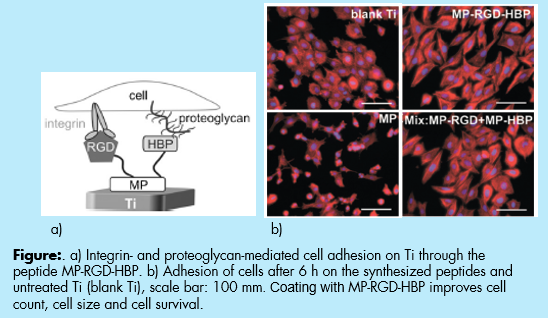

Peptide for Titanium and Biomaterial Surface Coating for Enhanced Osseointegration
Patents for licensing Biomedicine and Biotechnology Center - Leipzig University

Microbial DNA Sequencing-Microbialtec Research
Research Services and Capabilities Microbialtec Division

Heat2Power – Plant-scale optimisation of electricity production from waste heat
Innovative Products and Technologies Luxembourg Institute of Science and Technology (LIST)

Antibody Based Therapy to Treat Cancer and GI Related Diseases
Patents for licensing Yeda

Modelling Cystic Fribrosis and measuring microbial stickiness
Knowhow and Research output National Biofilms Innovation Centre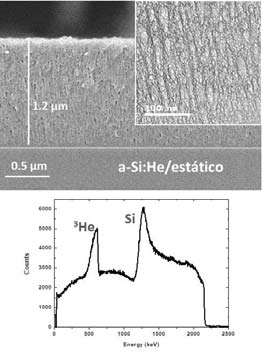

Production of porous solids with embedded gases to be used as solid-gas nanocomposite materials
Patents for licensing Consejo Superior de Investigaciones Científicas

A bioprocess aiming at specific micropollutants reduction
Innovative Products and Technologies Luxembourg Institute of Science and Technology (LIST)

New method for rapid, simple and efficient detection of nitrites
Patents for licensing Universidad de Alicante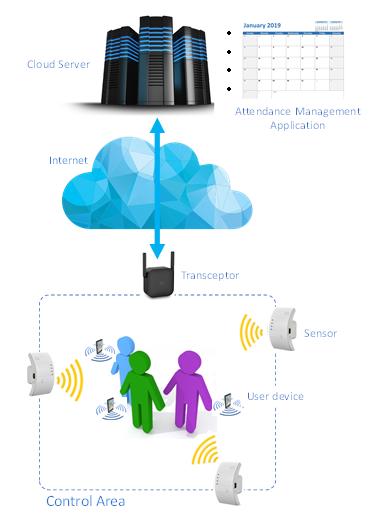

Innovative unattended presence control system for workers
Patents for licensing Universidad de Alicante

Hidracot, Organic Matter Removal (CatalyticAdvanced Oxidation, COD < 5.000 ppm)
Patents for licensing Fundació URV

APTAMER : Small single-stranded nucleic acid (DNA or RNA) for virus detection
Innovative Products and Technologies Luxembourg Institute of Science and Technology (LIST)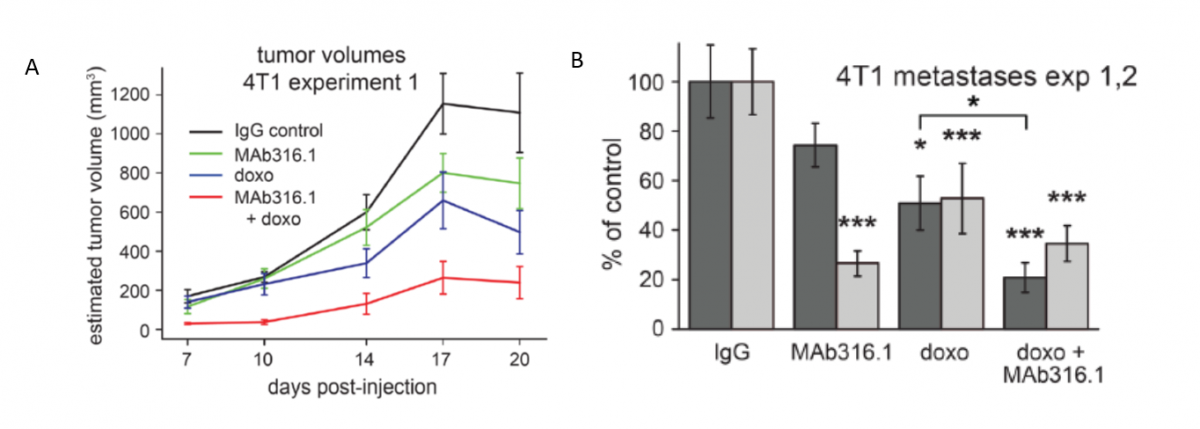

Anti-QSOX1 Antibody for Treating Cancer and Metastasis
Patents for licensing Yeda

Sustainable siderurgic/recycled-aggregate high-performance roller-compacted concrete
Patents for licensing UNIVERSIDAD DE BURGOS

