Showing 1 to 15 of 2075 results


CHEMIOTHERAPY: IRINOTECAN DRUG MONITORING
Patents for licensing National Cancer Institute CRO Aviano

Nitroxoline for prevention and treatment of coronavirus disease
Patents for licensing University of Ljubljana

Soy-Based Paste Wax to Replace Carnauba
Innovative Products and Technologies Airable Research Lab, business line of Ohio Soybean Council

Optimized analysis of slurry in agriculture
Patents for licensing Unismart - University of Padua Foundation

Textile Reinforced Concrete Prototyping Technology
Knowhow and Research output Steinbeis Centre for Technology Transfer India

Market research, consumer studies and advice for the development of online and mobile commerce
Research Services and Capabilities UNIVERSIDAD DE BURGOS

Prefabricated of cement with fire resistant polymeric residues
Patents for licensing UNIVERSIDAD DE BURGOS

Monitoring of Esca viticulture disease symptoms
Innovative Products and Technologies Luxembourg Institute of Science and Technology (LIST)

Graphene-based electroactive fluids for Energy Storage
Patents for licensing Consejo Superior de Investigaciones Científicas

Surface Plasmon Amplification of stimulated emission
Knowhow and Research output RAMOT at Tel Aviv University Ltd.![In vitro method for detecting tumor growth and diagnosing or prognosticating the risk of metastasis in a human subject that has be[…]](https://static8.innoget.com/uploads//2db5331b60422d5d9eb1fc1c6584193c3db79682.png)

In vitro method for detecting tumor growth and diagnosing or prognosticating the risk of metastasis in a human subject that has be[…]
Patents for licensing Fundación Instituto de Investigación Sanitaria de Santiago de Compostela (FIDIS)

Catalyst for synthesis gas (syngas) production using carbon dioxide (CO2) and
Patents for licensing CSIC - Consejo Superior de Investigaciones Científicas

Fast detection of microorganisms using microcalorimetry
Patents for licensing University of Vigo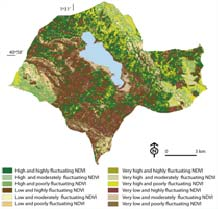

Environmental indices through LandSAT images
Patents for licensing Consejo Superior de Investigaciones Científicas

