Showing 1 to 15 of 2075 results


Microfluidics device for SERS
Patents for licensing Consejo Superior de Investigaciones Científicas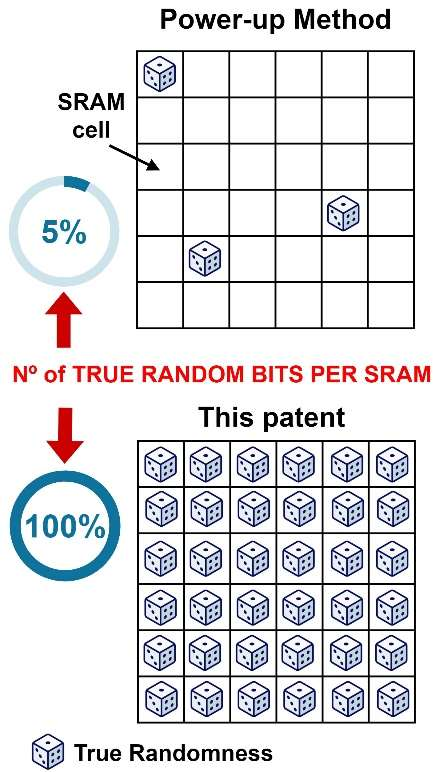

Method and device for generating true random numbers
Patents for licensing CSIC - Consejo Superior de Investigaciones Científicas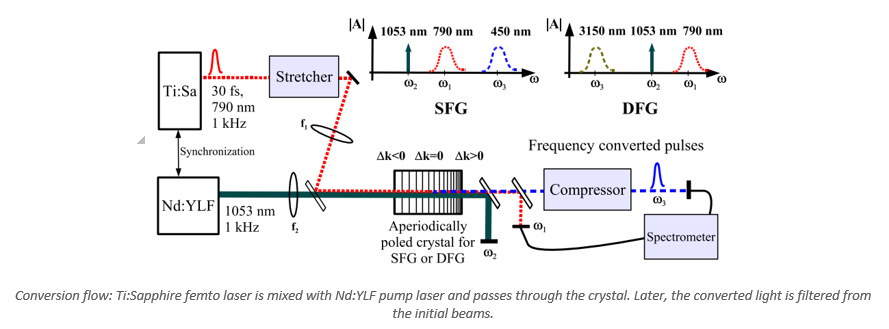

Optical Conversion of Ultra-Fast Laser Pulses
Patents for licensing Yeda

Bracelet for postural control
Patents for licensing UNIVERSIDAD DE BURGOS

HORECA: Platform for selection of employees for the service sector
Innovative Products and Technologies Fundació URV

Immobilization of glucose-oxidase for oenological application to obtain wines with low alcohol content
Research Services and Capabilities UNIVERSIDAD DE BURGOS

Quick RESCUE. New foldable, self-inflating, light and highly portable device for water rescue and lifesaving
Innovative Products and Technologies DIPSALUT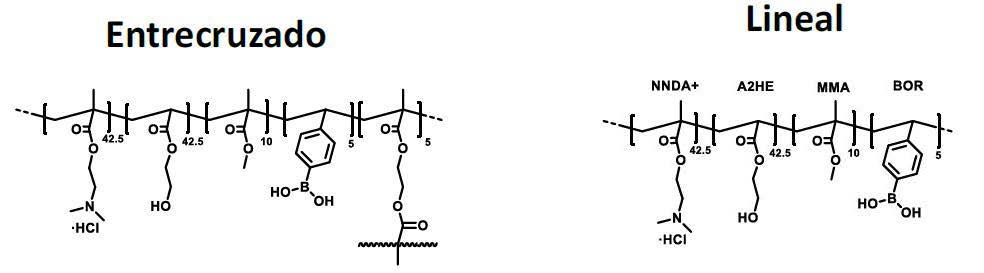

Polymer capable of extracting aromatic alcohols from honey
Patents for licensing UNIVERSIDAD DE BURGOS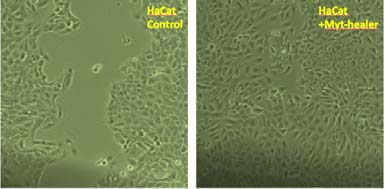

Myt-healer: a new potent wound healing peptide
Patents for licensing Consejo Superior de Investigaciones Científicas

ELECTRE-H software package: a tool for data analysis and decision aiding with hierarchical criteria
Innovative Products and Technologies Fundació URV

Odor Detection Platform - Low power, Low cost, Programmable Sensor Array
Innovative Products and Technologies University of Manitoba

Reinforced packaging paper
Patents for licensing CSIC - Consejo Superior de Investigaciones Científicas

Chimeric protein with high antimicrobial activity
Patents for licensing Consejo Superior de Investigaciones Científicas

Gas cylinders to have QR codes to stop theft
Innovative Products and Technologies XiangKang

