Showing 1 to 15 of 2075 results


Textile Reinforced Concrete Prototyping Technology
Knowhow and Research output Steinbeis Centre for Technology Transfer India

Nitroxoline for prevention and treatment of coronavirus disease
Patents for licensing University of Ljubljana

Soy-Based Shrinkage Reducing Additive
Innovative Products and Technologies Airable Research Lab, business line of Ohio Soybean Council

Quick RESCUE. New foldable, self-inflating, light and highly portable device for water rescue and lifesaving
Innovative Products and Technologies DIPSALUT

Synthesis of Phase pure Titanium dioxide Nanoparticles
Knowhow and Research output Steinbeis Centre for Technology Transfer India

Procedure for the determination of cocaine using an enzymatic biosensor
Patents for licensing UNIVERSIDAD DE BURGOS

Dispensing system for flexible packaging
Innovative Products and Technologies impulse

Monitoring of Esca viticulture disease symptoms
Innovative Products and Technologies Luxembourg Institute of Science and Technology (LIST)

Device to determine the food shelf-life
Patents for licensing Consejo Superior de Investigaciones Científicas

Immunotherapy for Stroke and Cognition in a Cerebral Amyloid Angiopathy Animal Model
Knowhow and Research output RAMOT at Tel Aviv University Ltd.

Process to produce lithium carbonate from spodumene ore concentrates.
Patents for licensing Universidad de Concepción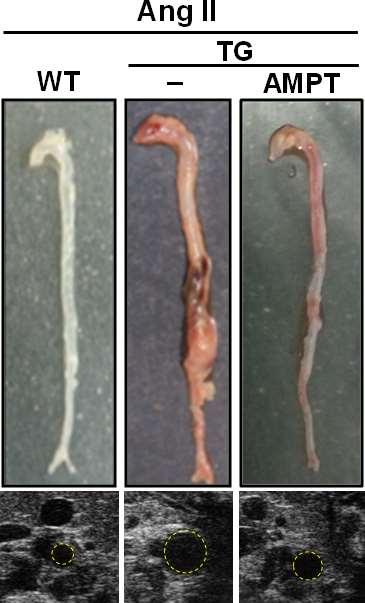

Tyrosine hydroxylase inhibitors for treatment of aortic aneurysm
Patents for licensing CSIC - Consejo Superior de Investigaciones Científicas

Innovative process for the removal of persistent organic pollutants present in wastewater streams
Patents for licensing University of Vigo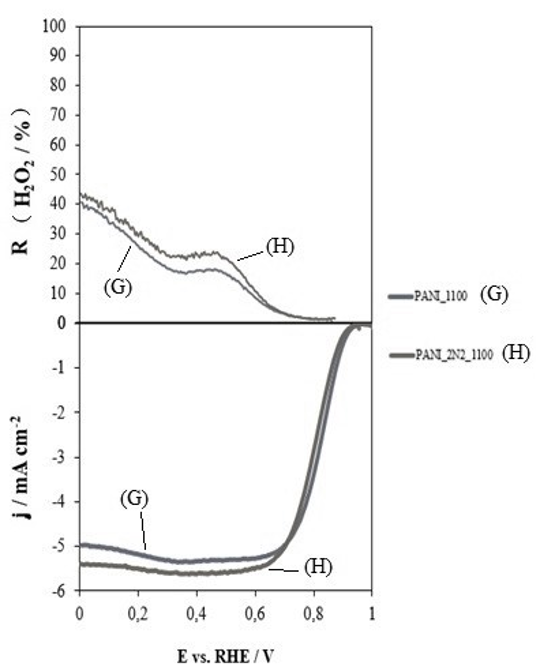

New metal-free electrocatalysts for fuel cells
Patents for licensing Universidad de Alicante

