Showing 1 to 15 of 2075 results


Aortic aneurysm treatment device
Patents for licensing IBIMA-Plataforma Bionand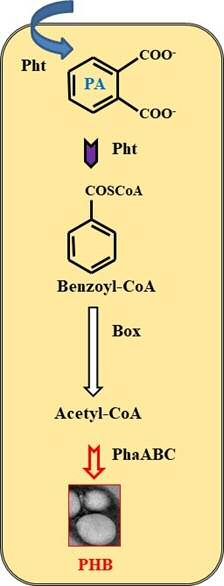

Two in one: engineering recombinant biocatalysts for plastic recycling and towards bioplastic production
Patents for licensing Consejo Superior de Investigaciones Científicas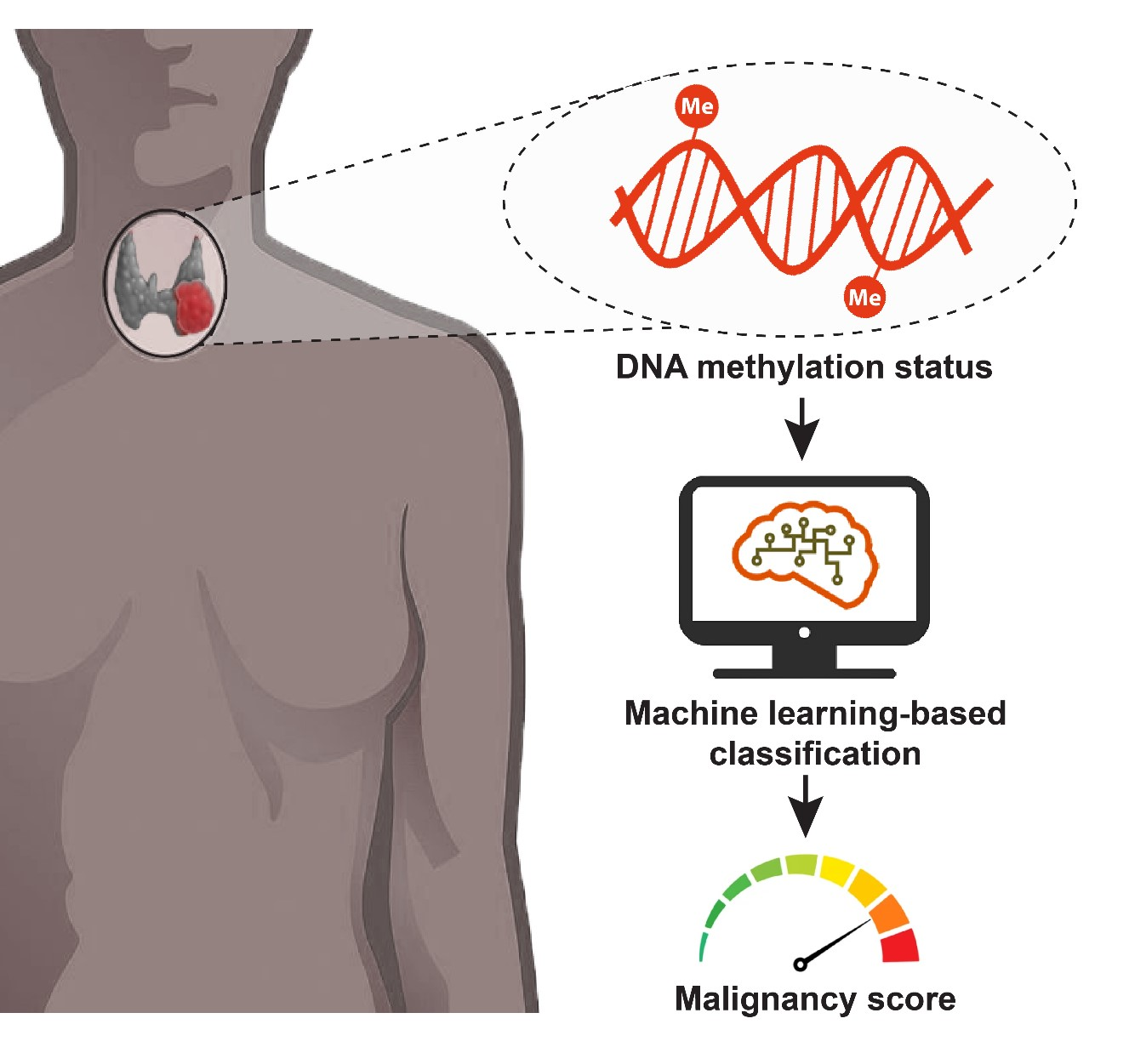

Method for the diagnosis of thyroid cancer based on epigenetic biomarkers
Patents for licensing CSIC - Consejo Superior de Investigaciones Científicas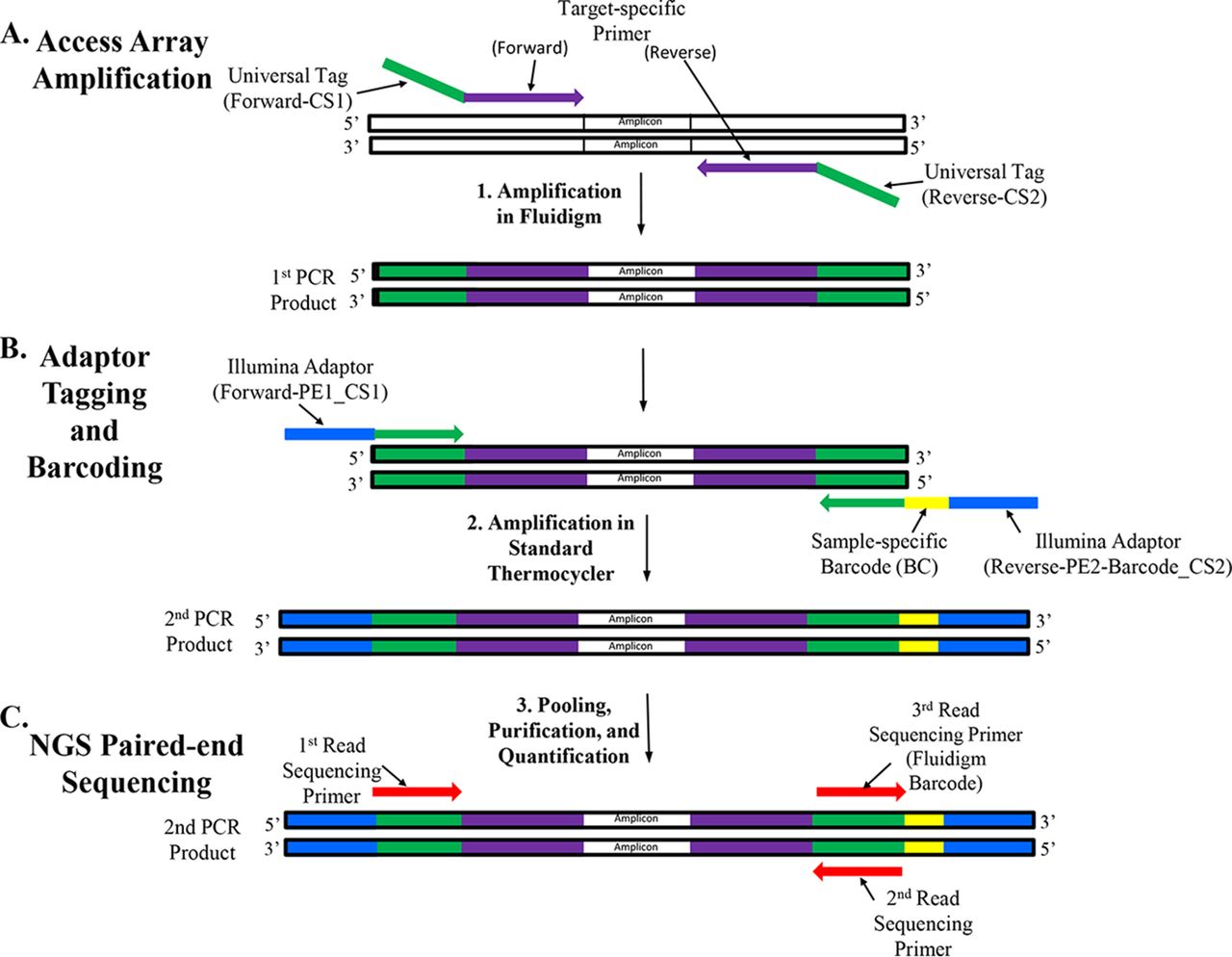

Amplicon Sequencing-Microbialtec Research
Research Services and Capabilities Microbialtec Division

Method for preparing advanced Eco-friendly incombustible biocomposite
Innovative Products and Technologies Korea Institute of Energy Research

Heat2Power – Plant-scale optimisation of electricity production from waste heat
Innovative Products and Technologies Luxembourg Institute of Science and Technology (LIST)

3D-printed anaerobic reactors
Knowhow and Research output National Biofilms Innovation Centre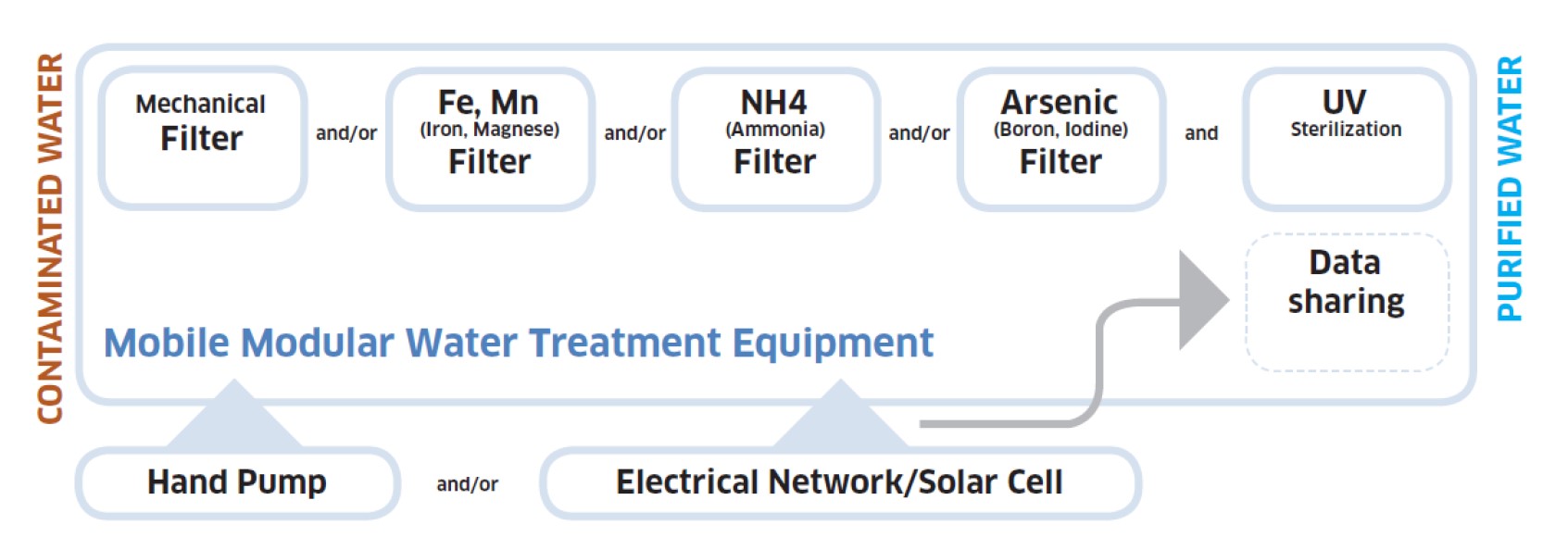

Chemical Free Regenerable Water Filtering System for Small Communities with Contaminated Water
Innovative Products and Technologies Laser Consult Ltd.

Non-invasive method for the diagnosis and screening of colorectal cancer and advanced adenomas in asymptomatic individuals
Patents for licensing CINBIO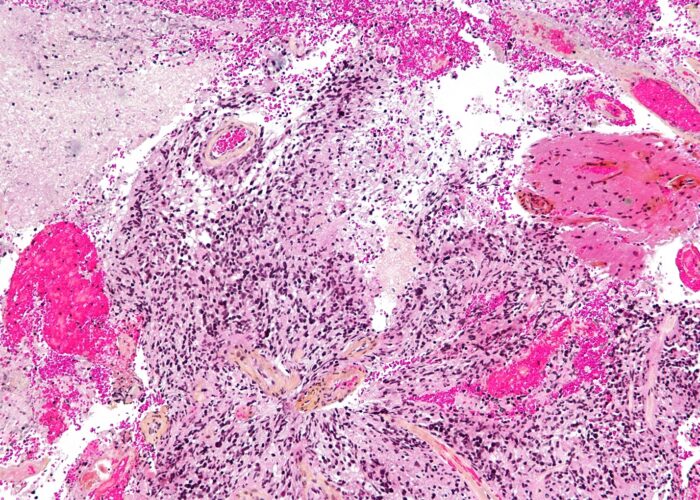

COMPOSITION FOR USE IN THE TREATMENT OF GLIOMA AND GLIOMA-INDUCED EPILEPSY
Patents for licensing SISSA

Laboratory for Trace Organic Analyses - Cracow Univerisity of Technology
Research Services and Capabilities Cracow University of Technology

Preparation method of palladium alloy composite membrane for hydrogen separation.
Innovative Products and Technologies Korea Institute of Energy Research

New quinoline compounds for use in the treatment and prevention of viral
Patents for licensing CSIC - Consejo Superior de Investigaciones Científicas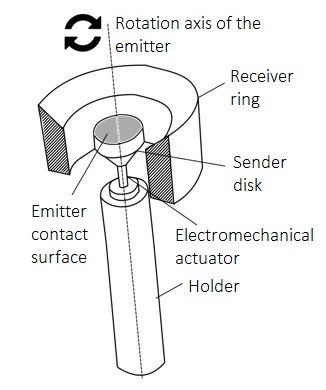

Device for measuring the biomechanical properties of the cornea
Patents for licensing Universidad de Granada

