Showing 1 to 15 of 2075 results


Predictive lab models of the human digestive and respiratory systems.
Innovative Products and Technologies National Biofilms Innovation Centre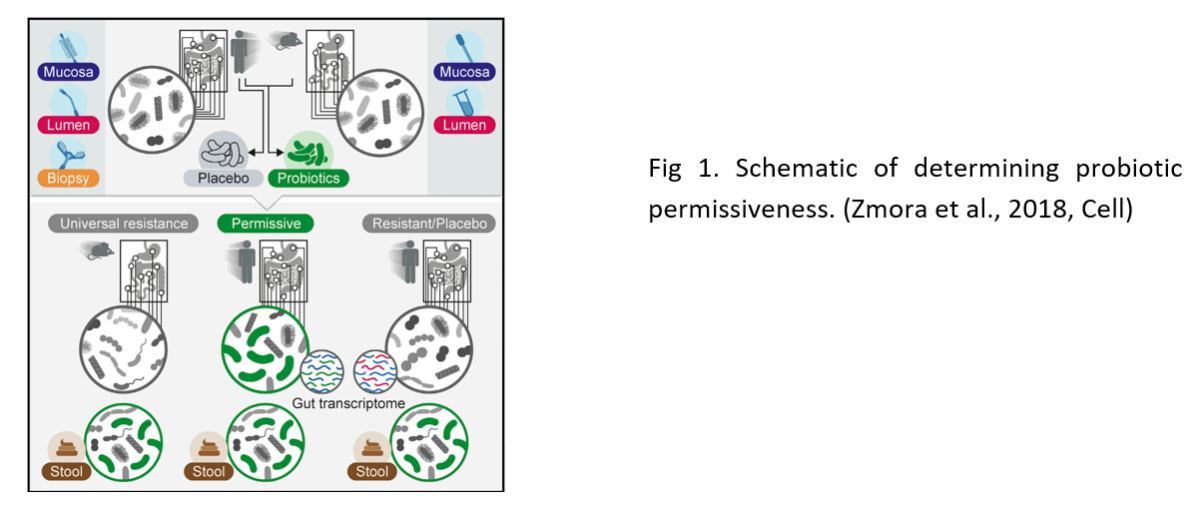

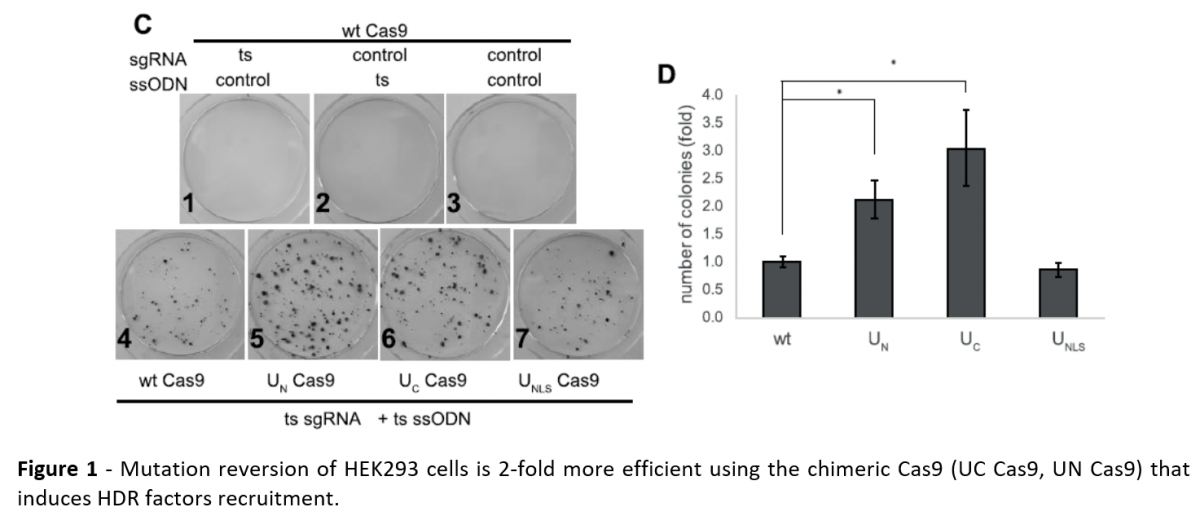
Person-specific Assessment of Probiotics Responsiveness
Patents for licensing Yeda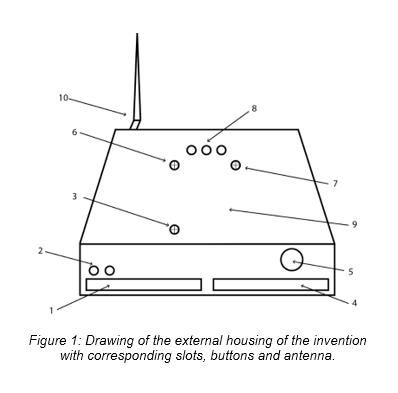

Seismic-noise recording system with real-time data visualisation
Patents for licensing Universidad de Alicante

AONIR PLATFORM, for in line and real-time production characterization based on NIRS technology.
Innovative Products and Technologies AOTECH![Nanometric markers: High-efficacy and fast molecular finding Authors: Luis Liz Marzán, Ramón Álvarez Puebla and Javier García […]](https://static5.innoget.com/uploads//a624a02c650d37812ff90bde0ebe9b04dfa7aa38.png)

Nanometric markers: High-efficacy and fast molecular finding Authors: Luis Liz Marzán, Ramón Álvarez Puebla and Javier García […]
Patents for licensing University of Vigo

Correction and monitoring of laser phase noise in long-haul coherent optical multicarrier systems
Patents for licensing Instituto de Telecomunicações

Enzyme products for your industrial reactions
Innovative Products and Technologies Zymvol Biomodeling S.L

SolveAir UNI - air disinfectant device
Innovative Products and Technologies Laser Consult Ltd.![Furalkamine - new class oform-control form-control f phenol-free mannich base curing agents for epoxy resins; offering excellent […]](https://static3.innoget.com/images/bgpremium.png)

Furalkamine - new class oform-control form-control f phenol-free mannich base curing agents for epoxy resins; offering excellent […]
Innovative Products and Technologies Bitrez Limited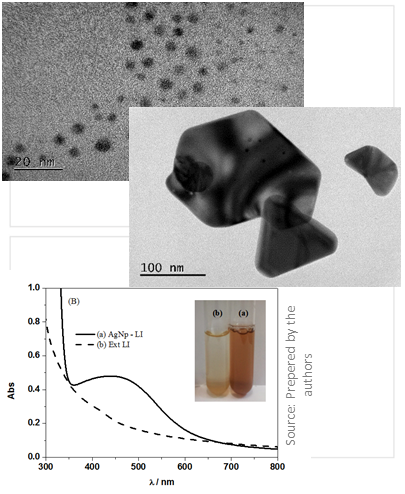

PROCESS TO OBTAIN SILVER NANOPARTICLES, SILVER NANOPARTICLES AND THEIR USES
Patents for licensing USP Innovation Agency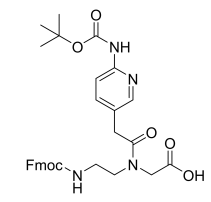

Fmoc-PNA-M(Boc)-OH Cas 1417611-27-8
Innovative Products and Technologies Creative Peptides

Aquatic platform - Luxembourg Centre for Systems Biomedicine
Research Services and Capabilities University of Luxembourg

Ultrafast quantum random number generator Authors: Marcos Curty, Valerio Pruneri, Morgan Mitchell and Marc Jofre
Patents for licensing University of Vigo

N4 -acyl-2-deoxycytidine-5-triphosphates for the enzymatic synthesis of modified DNA
Patents for licensing Vilnius University