Showing 1 to 15 of 2075 results

Selective estrogen receptor modulators-Like Library
Innovative Products and Technologies Otava Research Institute

QUOSCIENT - Fully automated cybersecurity platform to manage operative processes
Innovative Products and Technologies EIT Digital

Veggletto Vegetarian Egg Replacement Technology
Patents for licensing Byron Food Science Pty Limited

An Off-Line Image Search Engine
Patents for licensing Yeda

Advanced electro catalyst for the batteries
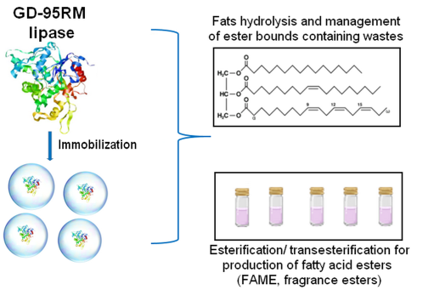
Innovative Products and Technologies Nu innovations Inc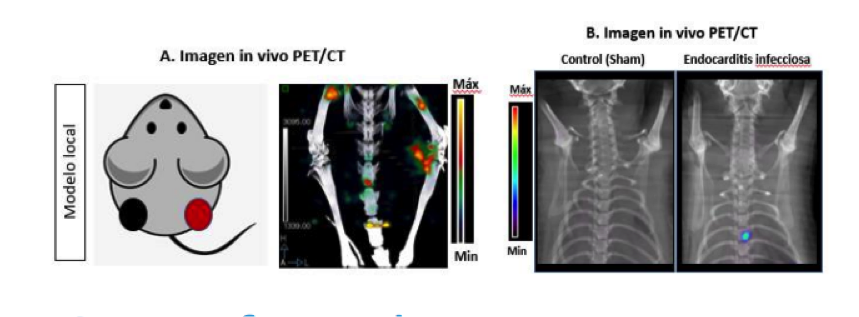

Radioactive compounds for non-invasive detection of Grampositive infectious diseases
Innovative Products and Technologies Instituto de investigación Sanitaria Gregorio Marañon (IiSGM)

JUSTSNAP - Turning Retail Receipts into Ecommerce Billboards
Innovative Products and Technologies EIT Digital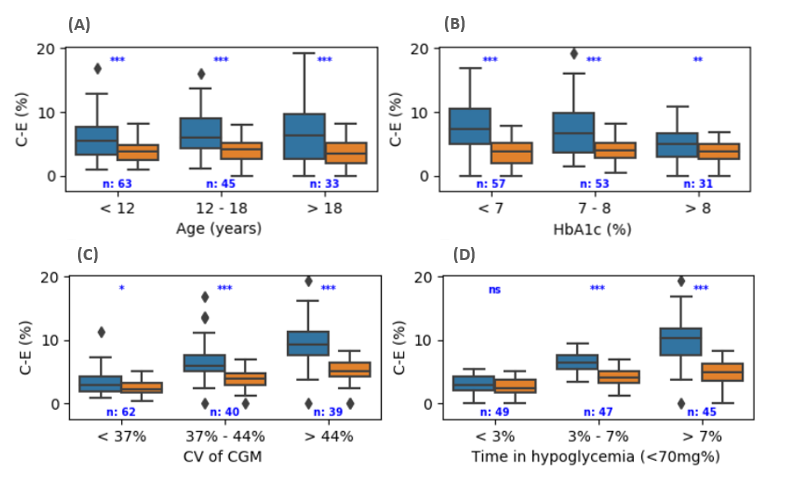

Artificial Pancreas
Patents for licensing Yeda

ARCH·E10 is a small, fast and efficient solution for recording archaeological and palaeontological data during fieldworks.
Innovative Products and Technologies Fundació URV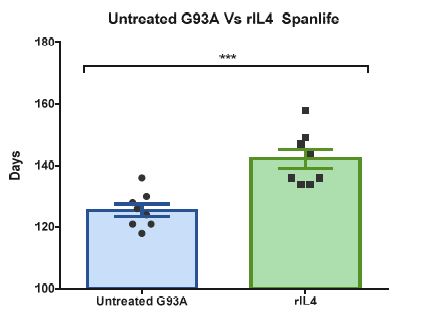

Treatment and prevention of Amyotrophic Lateral Sclerosis
Patents for licensing Universitat de Lleida

SYNERISE - Next Generation AI Marketing Cloud
Innovative Products and Technologies EIT Digital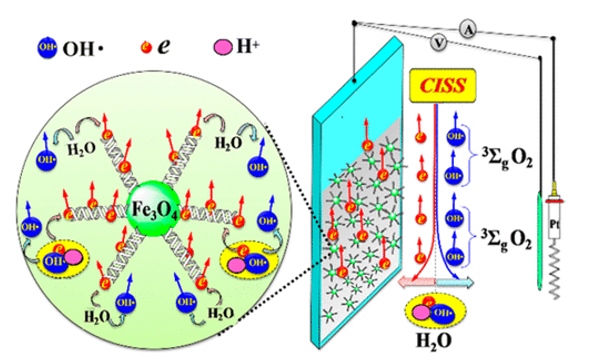

Spin Controlled Water Electrolysis for Efficient Production of Hydrogen
Patents for licensing Yeda

Manufacture of an eco-cobble from recycled material
Patents for licensing UNIVERSIDAD DE BURGOS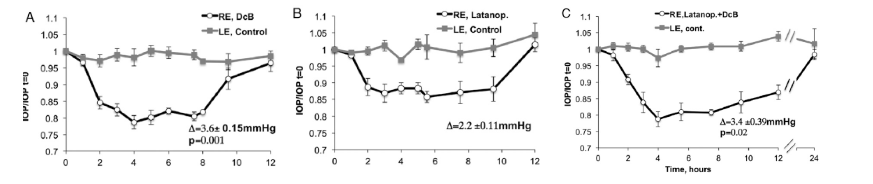

Improved Treatment for Glaucoma
Patents for licensing Yeda